Note that data could have been collected with th… Which of the following thermometers can respond quickly to small temperature changes and are used in small or precise locations? In such gas thermometers, gas temperature changes in the bottle are proportional to pressure changes. (c) List four advantages and four disadvantages of mercury as thermometric liquid.Metrologists (experts in the science of measurement) have chosen the constant-volume gas thermometer for this purpose. (b) State three desirable properties of a thermometric liquid. (a) State three physical properties of substances which may be used to measure temperature.State one advantage which a constant-volume gas thermometer has over other thermometers and one reason why it is seldom used as an everyday laboratory instrument.(b) Give the reasons for the following features of the thermometer.(a) Draw a labeled diagram of a clinical thermometer and explain how it works.Describe the action of resistance thermometer.GENERAL EVALUATION ( Post your answer in the comment box for rating and discussion) The working principle of a thermocouple depends on the variation of the electromotive force (e.m.f) between junctions. The electrical resistance of the element in a platinum resistance thermometer at 100 0C, 0 0C and room temperature are 75.000Ω, 63.000Ω and 64.992Ω respectively. The temperature can be calculated using the equation below. The ends of the wire are connected to a wheatstone bridge which measures the resistance at 0 0C, at 100 0C and the resistance at the desired temperature. The higher the temperature the greater the resistance. This thermometer depends on the variation in the electrical resistance of a conductor with temperature. A constant volume gas thermometer records pressure of a body as 300mm of Hg at 0 0C and 400mmHg at 100 0 Calculate the temperature of the body when the gas pressure reads 350mmHg.Calculate the temperature of the body when the gas pressure reads 300mmHg. The pressure at 100 0C is also gotten by placing it over stem.Ī constant volume gas thermometer records pressure of a body as 250mm of Hg at 0 0C and 350mmHg at 100 0C. To use the thermometer it must be calibrated by getting the gas pressure at 0 0C, when the bulb containing is placed in pure melting ice. P=h+H, where H is the atmospheric pressure.īut when the level of mercury is lower, then When the level of mercury in A is higher than that of D, then The pressure of gas is read from the manometer. The right side of AC of the manometer is moved up and down in order to bring the mercury level on the other side to its original position D to ensure that the volume of gas is constant. On heating the bulb to a specific temperature, the gas expands and pushes the mercury down to tube A and consequently mercury level at C rises. The constant – volume gas thermometer depends on the variation in the pressure of a gas at constant volume with changes in the temperature of the gas. List four advantages of mercury over alcohol as a thermometric thermometer.State three desirable properties of a thermometric liquid.The lower end of Y indicates the minimum temperature. When the temperature falls the alcohol contracts and moves in anticlockwise direction, the mercury pushes steel index Y up. The maximum temperature is read from the lower end of x. When the temperature rises, since alcohol expands more than mercury, The alcohol expands and flows in clockwise direction and the mercury pushes steel index x upwards. It contains alcohol at two upper part of the bent tube and mercury below. (iii) The Maximum and minimum thermometer is used to measure the maximum and minimum temperatures of the day. (ii) The school thermometer is used in school laboratory, it ranges from 0 0C – 100 0C.
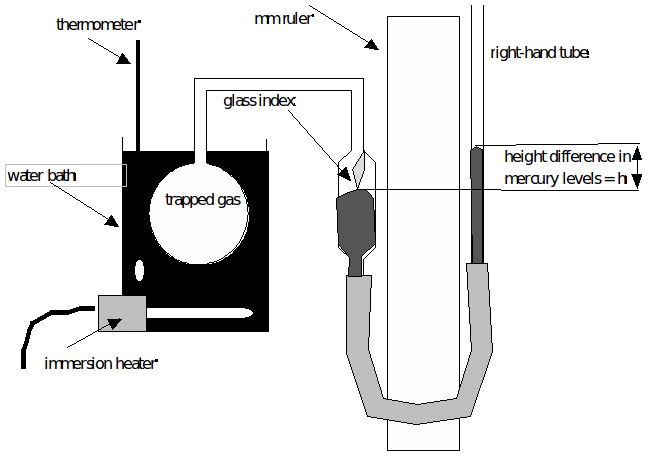

The narrow constriction prevents the mercury from flowing back into the bulb immediately after the thermometer has been removed from the patient’s body. (i) The clinical thermometer has a constriction and it has a short range (35 0C – 43 0C). The liquid-in-glass consists of the school thermometer, clinical thermometer, maximum and minimum thermometer. Any liquid that will be used as a thermometric liquid must be a good conductor of heat, be easily seen in glass, have a high boiling point, have a low freezing point, have a low specific heat capacity, must not wet glass and must expand uniformly. In which the change in volume of the liquid measures the change in temperature. The liquid-in-glass thermometer uses liquid(mercury) as thermometric substance.
